The HIV_Sequence Dataflow Description
Below we describe (high level) the HIV_Sequence dataflow between the data provider and the healthdata.be platform.
(source: DPD 01/2024)
Process A:
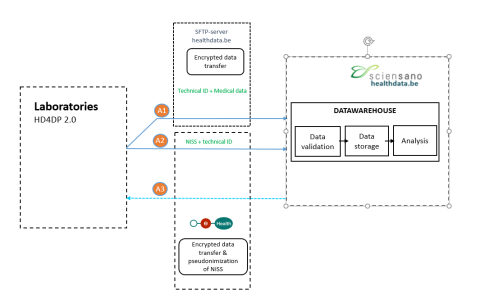
Data from laboratories will be collected via HD4DP 2.0 and transferred to healthdata.be in 2 separate flows:
Flow A1 containing medical data with an encrypted technical ID. The data is transferred, via SFTP, to HD-DWH. Because this file doesn’t contain data that needs to be pseudonymized the file will be directly send from the data providers to the HD-DWH by using SFTP.
Flow A2 containing the INSS of the patient with the same technical ID. The data is transferred, via eHealth (for encoding of the patient identification data by eHBox codage), towards the healthdata.be data warehouse (HD-DWH).
The technical ID is encrypted by the sender whereas the INSS is coded by the eHealth, as trusted third party. Upon receipt, the healthdata.be platform consolidates the separate communications based on the technical ID. After consolidation and technical quality control, the technical ID is immediately and permanently removed from the healthdata.be infrastructure. The healthdata.be platform will keep a log of these technical processes.
Via flow A3, data validation requests can be sent to data providers.
Data Users will have access to the HD DWH, according to the mandate received, so data validation and exploitation can take place.
The HIV virus nucleotide sequence along with a random, temporary technical identifier will be exported from the DWH environment and uploaded to the HIV Sequence Analysis program hosted by Stanford University (https://hivdb.stanford.edu/hivalg/by-sequences/). The result of this algorithm will be uploaded back to the HIV analysis environment to be associated with other variables for analysis (based on the random and temporary technical identifier). This information is necessary to analyze the epidemiology of antiretroviral drug resistance based on the characteristics of HIV-positive individuals. To this end, HIV sequences collected in AIDS reference laboratories should be interpreted in terms of the presence of resistance.
This export/import of sequence data from healthdata.be secure data warehouse will be regulated by a standard operating procedure which allows audits regarding the restrictive nature of the import/export.
Figure 1: Data flow used for HIV Sequence registry
 docs.healthdata.be
docs.healthdata.be